Internal Assessment for 2011 International Review - CIHR Institute of Genetics
Table of Contents
- Mandate and Context
- Institute Priorities
- Key Initiatives
- Initiative 1: Integrating the physical and applied sciences into health research – Novel approaches to biomedical research
- Initiative 2: From genes to genomic medicine, including clinical genetics research – Building a bridge between basic and clinical research
- Initiative 3: Population genetics, genetic epidemiology and complex diseases – Fostering community leadership
- Initiative 4: Health services for genetic diseases – Building a knowledge translation community for our genetic and biochemical discoveries
- Initiative 5: Genetic ethical, legal and social issues – Building more leaders and engaging the public
- Initiative 6: Enabling Canadian International Leadership
- Initiative 7: Establishing national meetings
- Outputs and Outcomes
- Going Forward
- List of Acronyms and Abbreviations
- References
List of Figures
- Figure 1: Percentage of total CIHR expenditures related to Institute of Genetics mandate for fiscal years 2000–2001 to 2009–2010
- Figure 2: Expenditures and number of grants related to the Institute of Genetics mandate for 2000–2001 to 2009–2010
- Figure 3: Specialization index and average of relative citations for top 10 countries publishing within the Institute of Genetics mandate, 2000–2008
Mandate and Context
The Institute of Genetics (IG) mandate is to support research on the human genome and on all aspects of genetics related to human health and disease, including how information contained in the genome is modified or altered through interactions with physical and social environments. Broad stakeholder consultations soon after the founding of CIHR indicated a clear need for IG to expand the interpretation of its mandate to recognize the implicit inclusion of basic biochemistry, cell biology and research using model organisms.
The Institute accepted this challenge. Today, IG is generally viewed as the institute that advocates for fundamental research using animal and cell culture models aimed at gaining basic insights into biological processes of relevance to human health. The IG has also demonstrated a strong commitment to knowledge translation (KT) and to research concerning ethical and health-service delivery issues that result from the genetics revolution.
Therefore, the present mission is to support research on the human and model genomes and on all aspects of genetics, basic biochemistry and cell biology related to health and disease, including the translation of knowledge into health policy and practice and the societal implications of genetic discoveries.
Canada is recognized internationally as a leader in the areas of genomics, genetics, biochemistry, cell biology and developmental biology. Among the many prominent members of the IG research community are Dr. Nahum Sonenberg, a premier international figure in research on the mechanism of protein synthesis and how it is controlled; Dr. Thomas Hudson, a leader in genomics research who was prominent in the international HapMap project; and Dr. Janet Rossant, an international leader in mouse developmental genetics.
While continuing to support and further strengthen our broad community, IG strives to:
- bring together strong research communities to work in new and exciting ways not possible without cross-collaboration
- nurture international research leaders in critical emerging areas
- foster the development of a community that will facilitate the knowledge transfer of genetic and biochemical discoveries
Institute structure and operations
IG underwent transition in early 2010 when the term of the inaugural scientific director, Dr. Roderick McInnes, was completed – the longest term of the 13 inaugural scientific directors. After a national search, Dr. Paul Lasko assumed leadership of IG effective May 2010. Dr. Lasko, a James McGill Professor at McGill University, is a developmental geneticist who studies post-transcriptional gene regulation in the Drosophila model organism. His appointment underscores IG's commitment to recognizing the importance of fundamental, discovery-based research in seeding the next generation of translational work. Dr. Lasko has also been active in genomics research, and he will build on his extensive previous work with the Human Frontiers of Science Program Organization to advance the IG's international agenda.
The Institute is guided in its operations by an Institute Advisory Board (IAB), composed of research leaders and partners with exceptional qualifications from Canada and abroad. In addition, the Institute's priority and planning committees, which enable more extensive community involvement than an IAB alone, are unique among CIHR institutes and were identified as a best practice in the previous International Review. Through these committees, members of the extended research community advise the scientific director and the IAB on strategic initiatives that need to be undertaken to implement the IG strategic plan effectively.
CIHR 2006 International Review observations
The 2006 CIHR International Review was largely supportive of IG and recommended that the Institute:
- sustain its efforts in the areas of research excellence, capacity development and funding strategic priorities
- maintain oversight and planning mechanisms, as they are effective
- sustain productive efforts in the area of ethics
- persist in its proactive efforts to be the voice of the basic sciences pipeline and to court leveraging opportunities
The report also recognized a continuing need for outreach to the basic biomedical science community, which would raise awareness and increase engagement in Institute activities, and to expand and make its communications efforts more effective. Other areas for ongoing development include raising the profile of IG's knowledge translation initiatives and monitoring the performance of its programs. The CIHR International Review report provides clear evidence that IG has demonstrated its commitment to continual improvement by addressing these recommendations – for example, implementing a newsletter and an end-of-grant report system.
Institute Priorities
Consulting with our community
The Institute has always been directed in a bottom-up manner, and its strategic directions have been set only after extensive consultations with its various stakeholders. For example, a national dialogue informed and refined the Institute's draft strategic plan. This plan was further modified at a strategic planning retreat held in September 2002, which was attended by approximately 60 leading researchers, academics, government and scientific policy makers in the genetics, biochemistry, cell biology and social science and humanities research communities. This meeting helped shape and finalize IG's first strategic plan.
To ensure that the strategic plan remained forward driven, IG dedicated its January 2008 meeting of the Institute Advisory Board (IAB) to strategic planning for the next five years. This expanded IAB meeting included all of the planning and priorities committee chairs and other research leaders working within the IG mandate. We discussed how to refocus and consolidate our activities in the context of the recommendations of the 2006 International Review that all of CIHR needs "to do less, better". This statement refers to the sense that CIHR was posting too many funding opportunities, and too many of them were of insufficient impact. We also examined whether our activities align well with overall Canadian science and technology strategy, the working vision for the renewal of CIHR's strategic plan, the needs and interests of the Canadian research community and international research directions.
We reached a consensus that IG's direction remains appropriate and decided to commence our next strategic planning process under the direction of our new scientific director and after we receive results from the 2011 CIHR International Review.
Current goals and research priorities
Advancing research and building capacity
The Institute must continue to support our strong research communities in the basic sciences of genetics, biochemistry and cell biology, while fostering a broad strategic translational agenda. The following five research priorities reflect our strategic focus:
- Integrating the physical and applied sciences into health research, including computational biology
- From genes to genomic medicine, including clinical genetics research
- Population genetics, genetic epidemiology and complex diseases
- Health services for genetic diseases
- Genetics and ethical, legal and social issues
Supporting individual investigators and strengthening the IG research community
Throughout the consultative process, there was a consistent request that the Institute provide clear and unwavering support for investigator-initiated research and for CIHR's Open Operating Grants Program. Other key activities include, but are not limited to, sponsoring national meetings and workshops that facilitate peer networking and mentorship, supporting initiatives that involve the mentorship of new researchers, and increasing Canadian contribution and visibility in international health research initiatives.
Establishing strategic partnerships – an early engagement philosophy
The Institute will engage partners and other stakeholders early in the process of developing research funding programs and related activities. To do this, IG established the Voluntary Health Organizations (VHO) Working Group to serve as a critical link between the Institute and the Canadian VHO community. This working group has expanded and grown into the Canadian Genetic Coalition, whose mandate includes promoting research, assisting with KT and informing policy in areas such as genetic non-discrimination.
Facilitating the knowledge transfer of genetic and biochemical discoveries
The Institute recognizes the enormous disparity between the extent of fundamental knowledge in genetics and genomics and the highly imperfect level of awareness of this knowledge and its application to health care. The scope and breadth of the Institute's mandate and the need to reach out to a diverse group of stakeholders requires a multi-faceted KT strategy.
Key Initiatives
The following section outlines key activities and investments for each IG research priority, plus our efforts to support individual investigators. The outputs and outcomes resulting from our key initiatives are presented later.
Initiative 1: Integrating the physical and applied sciences into health research – Novel approaches to biomedical research
Technical advances, such as faster computers, microarrays, microfluidics, new imaging technologies and high-throughput methods have been transformative for biomedical research. These technical advances stemmed from work in the physical sciences and engineering. Consequently, research at the interface between the physical and life sciences has enormous potential to accelerate medical advances. The goals of this research priority are to:
- provide catalytic funding for research areas with clear potential for significant improvements in health research and clinical practice
- enhance collaboration between physical and applied scientists and health researchers
- achieve long-term sustainability of funding for integrated research
The Institute alone has invested almost $20 million to support this strategic initiative since 2002. We have actively supported this research priority by:
- co-leading the Regenerative Medicine and Nanomedicine Initiative (RMNI)
- offering an annual funding opportunity to foster the development of tools, techniques and devices
- organizing national workshops
Since its inception, RMNI has been closely aligned with the priorities of eight CIHR institutes and has been engaged with other granting agencies, government departments and numerous voluntary health organizations (VHOs). As of June 2010, RMNI has secured total funding support in excess of $84 million. The focus for RMNI research funding has been on multidisciplinary, team-based approaches and innovative high-risk/high-benefit projects that have the potential to make a significant health impact.
One RMNI research team supported by IG promises to make heart transplants available to more infants. Previous studies by Dr. Lori West (University of Alberta) showed that ABO blood type-mismatched heart transplants are possible in infants because their immune systems are immature. The protocol she developed has now been adopted around the world, allowing more infants to be transplanted. Despite this success, donor hearts for infants needing transplants remain scarce. The Institute is co-funding a team, led by Dr. West, to test the idea that exposing the young immune system to very tiny synthetic ABO substances will induce tolerance, thus expanding the time window when a mismatched transplant can be performed safely. The team includes chemists and nanoscientists who will design and produce ABO substances and attach them to implantable devices. These implants will be altered to make them optimal for modifying the immune system and to allow tracking. Finally, they will be tested in a piglet heart transplant model. This is an exciting and entirely novel multidisciplinary project with enormous clinical potential.
We recognize that a diverse array of tools, techniques and methodologies are currently employed in health research and are an integral part of hypothesis-driven projects. Accordingly, the invention and development of new research tools or techniques, or the improvement and application of existing ones, are often crucial and tightly linked to the enabling of scientific discovery. To date, we have supported 43 projects focused on tools and methods development. Two competitions were in partnership with RMNI and one with the Institute of Neurosciences, Mental Health and Addiction (INMHA).
Through our Integrating the Physical and Applied Sciences Planning and Priorities Committee, and in partnership with the Institute of Cancer Research (ICR), the Institute of Infection and Immunity (III) and INMHA, we have brought together physicists, chemists and engineers with biomedical researchers to identify emerging research trends and develop approaches that encourage effective interdisciplinary research and training. Each meeting produced a white paper outlining key recommendations for both CIHR and the National Sciences and Engineering Research Council (NSERC), including the important issue of sustainable funding for research at the CIHR-NSERC interface. In particular, the Science Convergence Initiative (SCI) emerged from the second meeting. The initiative is viewed as a bold and innovative action plan to support cutting-edge research at the interface between the physical and applied sciences on the one hand and biological sciences on the other.
Initiative 2: From genes to genomic medicine, including clinical genetics research – Building a bridge between basic and clinical research
The challenging, multi-step process leading from basic research discoveries to their ultimate integration into clinical practice requires the active participation and collaboration of clinical genetics investigators with many research disciplines and partners. It is a high priority of IG to facilitate the translation of modern genetic and genomic knowledge to medical practice. The goals of this research priority are to fund research that bridges clinical investigation with more basic scientific research while also building clinical investigator research capacity.
In an effort to foster the translation of gene discovery to the clinic, IG has led two programs to support teams designed to bridge basic and clinical research: Genomic Medicine and Human Development; and From Genes to Proteins, Cells, Tissues and Patients. Collectively, the investment by IG, other CIHR institutes and VHOs into these two programs represents a financial commitment of more than $30 million over five years.
The Genomic Medicine and Human Development program funds large operating grants for research that bridges clinical investigation and more basic scientific research that addresses an important problem in human development. Building on Canadian strengths in developmental genetics and clinical research, the more than $13.9 million investment supports five team-based research projects in partnership with Institute of Circulatory and Respiratory Health (ICRH), Institute of Gender and Health (IGH), Institute of Human Development, Child and Youth Health (IHDCYH) and the Heart and Stroke Foundation of Canada.
The From Genes to Proteins, Cells, Tissues and Patients funding program supports interdisciplinary research teams that bring together geneticists with researchers in other areas to study a gene or protein strongly implicated in a human genetic disease or inherited mouse disease phenotype. Fostering collaboration between these disciplines is essential to enhancing the translation of gene and protein research to medicine. In January 2010, more than $16 million in support of seven research teams was announced by IG in partnership with Institute of Aging (IA), ICR, ICRH, Institute of Musculoskeletal Health and Arthritis (IMHA), the Canadian Cystic Fibrosis Foundation, the Crohn's and Colitis Foundation of Canada, and the Foundation Fighting Blindness, Canada.
One exciting project funded in 2010 through this IG-led initiative involves a team led by Dr. Michael Hayden (University of British Columbia). Protein palmitoylation is a dynamically regulated posttranslational modification critical for proper cellular trafficking, and defects in palmitoylation of selected neuronal proteins are associated with Huntington's Disease (HD). Dr. Hayden's team has developed a novel proteomic technology that will enable them to identify most of the neuronal proteins showing altered palmitoylation in HD and to define the relationship between defects in palmitoylation of these proteins and HD. This work promises to inform the development of small molecule inhibitors that target enzymes involved in palmitoylation, which could provide a novel class of therapeutic tools for patients with this devastating disease for which there is currently no effective therapy.
The IG Clinical Investigatorship and the Scriver Family MD/PhD Studentship Awards are addressing the need to build Canadian capacity in clinical research. The Investigatorship program is unique in Canada, and gives early-career clinical investigators the opportunity to initiate and conduct research in clinical genetics by contributing to their salaries and freeing up research time. The IG has invested more than $4 million in this program in support of 15 awards since 2002. In a partner program that focuses on student trainees, IG, with the help of the Canadian Gene Cure Foundation (CGCF), has supported 23 Scriver Family MD/PhD Studentship Awards for a total investment of more than $3 million.
Initiative 3: Population genetics, genetic epidemiology and complex diseases – Fostering community leadership
The new frontier of population health in the next five to 10 years will be based in genetic epidemiological research. The Institute's objectives in this area are two-fold: to provide the analytic expertise and tools to identify and characterize genetic determinants for diseases, and to facilitate the proper use and integration of these genetic determinants in subsequent molecular biology experiments as well as clinical and public health interventions. In this way, IG hopes to contribute to the prevention of common, complex diseases. In advancing this research priority, we have built an especially close partnership with the Institute of Population and Public Health (IPPH). To achieve the priority's objectives, the goals of this strategic initiative are to:
- build capacity among a community of scientists skilled to bridge laboratory-based research and population-based research
- facilitate tools and methods development
- make better use of unique Canadian resources such as founder populations and a universal health care system
Innovative population genetic and genetic epidemiologic methods are needed to facilitate studies of complex genetic diseases. In response, IG and IPPH have invested more than $1.5 million in programs designed to foster the development of new theories, strategies and methodologies that will facilitate more conclusive, high-quality etiological studies of such diseases. The Institute and IPPH have made almost $1 million available in support of training awards for genetic epidemiology researchers since 2002. In addition, we have made significant efforts through our Population Genetics and Genetic Epidemiology Planning and Priority Committee to establish and organize the annual Canadian Statistical Genetics and Genetic Epidemiology Meeting in addition to other community building activities such as videoconference journal clubs. These activities have helped bring researchers together and have fostered the development of leaders in the community.
This increased leadership capacity enabled the community to respond and secure a Strategic Training Initiative in Health Research (STIHR) grant in 2009. The IG-funded Strategic Training for Advanced Genetic Epidemiology training program, based at the University of Toronto under the leadership of Drs. France Gagnon, Shelly Bull, Steven Narod, Andrew Paterson and Lie Sun, offers graduate and postdoctoral training at the interface between statistical, biomedical and human population health sciences with a focus on genetics and genomics. Students are trained to exercise leadership in scientific investigations aimed at understanding and addressing the etiology of common chronic and infectious diseases and improving associated population health outcomes.
Initiative 4: Health services for genetic diseases – Building a knowledge translation community for our genetic and biochemical discoveries
Health services and policy research in genetics is essential to generate the evidence that health care system and knowledge users require to integrate genetic discoveries into policy and practice appropriately. The primary goal of this research priority has been to build researcher and receptor capacity in Canada. Meanwhile, it addresses the concern that Canada, like many countries, has a mismatch between its advanced capacity to generate fundamental genetics knowledge and its lesser capacity to determine how the health care system uses that knowledge, and when it needs to use it.
In partnership with the Institute of Health Services and Policy Research (IHSPR), IG has invested almost $10 million in this area, primarily to support emerging teams and operating grants. A series of consultative workshops defined the strategic focus for the investments. The workshops included researchers, provincial and federal policy makers and other partners such as IPPH, ICR, the Canadian Agency for Drugs and Technologies in Health, the Heart and Stroke Foundation of Canada and the former Federal/Provincial/Territorial Coordinating Committee on Genetics in Health.
The Human Genome Project has raised great expectations for personalized medicine. However, research efforts that help work out the many thousands of possible gene-disease associations must be matched by evidence on how the Canadian health care system can use this knowledge. A recently funded IG and IHSPR emerging team led by Dr. Brenda Wilson (University of Ottawa) is examining two areas where genomics meets everyday health care and general populations: using family history information to predict risk of common diseases and screening newborns for rare but serious inborn metabolic diseases. The team is engaging with citizen and health professional groups to understand their reactions to upcoming developments in genomic profiling in which DNA is used to assess the susceptibility of adults and children to common diseases.
The IG and IHSPR Health Services for Genetic Diseases Planning and Priority Committee engaged in a groundbreaking discussion, subsequently published in Healthcare Policy (2008),Footnote 1 that argued in favour of specialized health services and policy research in genetics. In addition, the committee organized sessions at the Canadian Association of Health Services and Policy Research Annual Meetings (2006, 2008) to generate interest in the broader health services/policy community to explore research questions related to genetics.
Initiative 5: Genetic ethical, legal and social issues – Building more leaders and engaging the public
Appropriate uptake of genetic research is largely contingent on how end users perceive the risks and benefits associated with this rapidly expanding field. The goal of this research priority is to identify and manage systematic barriers to uptake and the effective implementation of genetic services.
Although Canada has many internationally respected leaders in genetic ethical, legal and social issues (GELS) research, IG believes that the rapid emergence of numerous novel ethical, legal and social issues associated with genetic and genomic research requires substantial additional capacity. Therefore, IG has invested $2.4 million to support training and research in partnership with CIHR's Ethics Office, IPPH and IHDCYH since 2002. One recently funded IG GELS strategic operating grant, led by Dr. Bartha Knoppers (McGill University), is focused on defining stakeholders' rights and obligations in population biobanks through Material Transfer Agreements.
Through its GELS Planning and Priority Committee, IG has worked with the CIHR Ethics Office toward coordinating the efforts of all Institute ethics designates. Nationally, this committee organized three highly successful symposia related to interdisciplinary research, the communication of research results and issues emerging with the advent of personal genomics (described in more detail in the next section of this report). Another example is the Where Genomics, Public Policy and Society Meet (GPS) program, launched by Genome Canada in 2009, where IG is a core advisory partner. The objective of GPS is to broker a two-way dialogue between federal policy makers and GELS researchers on various policy options for addressing issues that arise at the interface of genomics and society.
Initiative 6: Enabling Canadian International Leadership
Structural Genomics Consortium
Led by Canadian researcher Dr. Aled Edwards, a world-leading expert in structural biology, the Structural Genomics Consortium (SGC), based in Toronto, Oxford and Stockholm, involves more than 200 researchers and is the world's leader in high-throughput determination of the three-dimensional structures of proteins relevant to human health, including those associated with cancer, neurological disorders and infectious diseases such as malaria. Information produced by the SGC is released into the public domain, sparking hundreds of downstream projects that provide insight into the functions of these proteins and their role in either safeguarding health or increasing susceptibility to disease.
The SGC has produced more than 1,000 high-resolution protein structures and a similar number of specific chemical probes, all available for open access. It is a remarkable example of a successful multinational collaboration. It has raised more than $100 million from public sector and charitable sources as well as from several large pharmaceutical companies.
The Institute, through its scientific director, represents CIHR on the governing board of this consortium and thus helps support and guide the activities of the SGC. The SGC plans large-scale production of antibody reagents against specific proteins, placing a high priority on those involved in epigenetic tagging.
Initiative 7: Establishing national meetings
The Institute established an innovative and highly popular annual New Principal Investigators (PI) meeting in 2002. Targeting faculty within the first four years of their appointment at a Canadian university, the IG New PI meeting fosters the formation of peer networks and provides mentoring on grant and paper writing and running a laboratory. Recently, mock peer review grant panels have been introduced. Between 2002 and 2009, more than 800 new PIs participated. The meeting has attracted partnerships with INMHA, ICR, IHDCYH, INMD, IA and IMHA. Keynote speakers have included internationally prominent researchers such as Drs. Thomas Jessell (Columbia), Huda Zoghbi (Baylor) and Hal Dietz (Johns Hopkins). Examples of anonymous comments provided in the workshop evaluations include, "Please encourage other CIHR institutes to implement a similar workshop, or participate in this one!", "Overall, this has been a very positive experience. It came across that CIHR cares and wants us to be successful", and "The meeting gave me a great introduction to the scientific community in Canada. What a great welcome."
This meeting also spurred development of the popular IG New Principal Investigator Guidebook. This publication is intended for all researchers who write grant applications in health research (including basic biomedical research and clinical research) as well as in the social sciences and humanities. In addition, the IG Maud Menten New Principal Investigator Awards, which recognize and support the research excellence of new PIs, emerged as a complement program to the meeting.
The Institute is responsible for establishing and organizing several recurrent national meetings that support research, knowledge translation, networking and collaboration. These include the Canadian Developmental Biology Conference, the Canadian Genetic Epidemiology and Statistical Genetics Meeting, the Canadian Human Genetics Conference and the New Principal Investigators Meeting. Before IG was established there was no national meeting in developmental biology. Since 2002, five such meetings have been held in biennial years, each attended by 150–300 participants, including clinicians studying birth defects and numerous participants from outside Canada. Since 2008 this meeting has been co-sponsored by the American Society for Developmental Biology, which promotes it among its regional meetings.
A more recent example of a national meeting successfully established by IG was the Canadian Human Genetics Conference, which brings together Canadian scientists with a broad interest in human genetics and animal models of genetic diseases to discuss topics of current research interest. In partnership with the CGCF, IG was able to reinvent the small annual meeting of the former Canadian Genetic Diseases Network and opened it to the broader human genetics research community from a network members-only meeting.
Outputs and Outcomes
This section of the report illustrates the impact of the Institute through trend data, bibliometrics, program evaluations and illustrative examples. The Institute has undertaken an evaluation of all of its strategic funding programs. Data collected from the completed end-of-grant and end-of-awards reports is being used to inform the Institute about to what degree its funding has enabled research programs across Canada. This information will play an essential role in enabling IG's strategic planning. Through evaluation efforts to date, 134 nominated principal investigators (PI) from IG grants and award competitions have been surveyed by IG, with a response rate of 70% (n=94/134). The results of these evaluations are reflected in the appropriate sections below. The Institute is committed to the continued monitoring of its programs every year.
Advancing knowledge
The strength and vitality of the IG research community is evident from its funding profile over the past 10 years, which has steadily increased. This, in turn, has resulted in impressive growth in the number of publications with Canadian authors in IG-relevant areas.
Increasing levels of funding
As Figure 1 shows, the IG research community, including basic biochemistry and cell biology, consistently receives a high proportion of funding from the CIHR Open Operating Grant Program – between 35% and 40% of total expenditures. For open training award competitions, the percentage of total CIHR expenditures peaked in 2006–2007. However, the CIHR absolute dollar investment increased from $13.7 million in 2005–2006 to $15.0 million in 2009–2010 (data not shown). In 2008–2009, the proportion of strategic grant funding can be partially explained by 28 grants funded under the Strategic Training Initiative in Health Research (STIHR) having completed their six-year term with only 12 new programs being funded the following fiscal year.
Figure 1: Percentage of total CIHR expenditures related to Institute of Genetics mandate for fiscal years 2000–2001 to 2009–2010
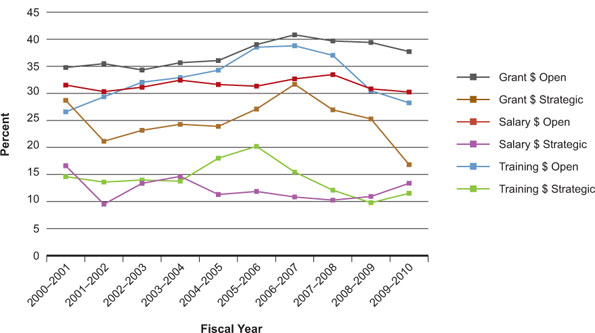
Importantly, as these figures are based on validated keyword search files, it is inevitable that there will be overlap between institutes as multiple counting occurs (for instance, a grant in the area of cancer genetics will be counted both for IG and for ICR). Although variable from year to year, grants are being counted from 1.7 times to 2.1 times, on average. This overstatement reflects the synergy and complementarity of our institutes and their mandates. Funding data are based on a keyword search of the CIHR funding database and validated through a subjective process. Projects may have multiple institute affiliation.
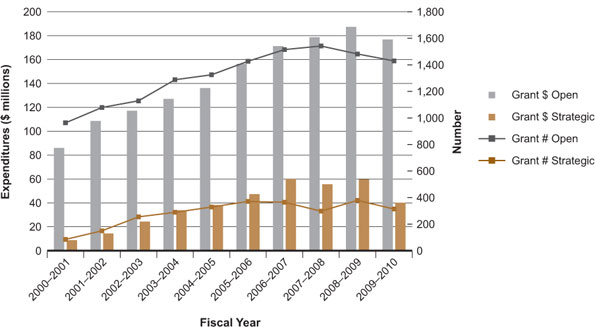
As shown in Figure 2, the IG research community was awarded $86 million through CIHR's Open Grant expenditures in 2000. Since then, there has been a remarkable upward trend, with investment increasing to $177 million in 2009–2010. Funding to IG researchers has been essentially stable since 2006–2007, reflecting the relatively static funding envelope for CIHR. However, the IG research community has capitalized effectively on earlier increases to the CIHR budget.
Figure 2: Expenditures and number of grants related to the Institute of Genetics mandate for 2000–2001 to 2009–2010
Increasing numbers of publications
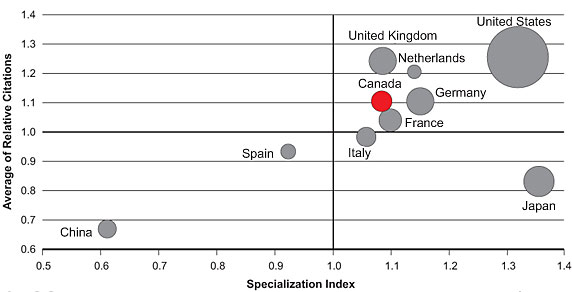
Figure 3 shows the average of relative citations (ARC) and specialization index (SI) for publications in IG mandate areas among the top 10 countries between 2000 and 2008. Canada scores above the world average (1.0) on both indices ranking fourth and sixth worldwide for ARC (1.10) and SI (1.08), respectively.
Between 2000 and 2008, both ARC and SI for Canadian publications show a general upwards trend from 1.10 to 1.18 for ARC and 1.06 to 1.08 for SI (data not shown). With 53,505, Canada ranks sixth worldwide for number of publications (as represented by the size of the circle), increasing annually from 4,828 in 2000 to 6,986 in 2008 (data not shown). These data indicate growth in number of publications, increasing citations of Canadian papers (ARC) as well as increasing specialization of Canadian research in IG mandate areas (SI).
Publications on genetics were identified through Medical Subject Headings (MeSH) searches done by the Observatoire des Sciences et des Technologies (OST). Databases searched may not cover all publications in this area and ARC data is incomplete for 2008. Countries were ranked based on total number of publications (2000–2008).
Figure 3: Specialization index and average of relative citations for top 10 countries publishing within the Institute of Genetics mandate, 2000–2008
Of the top 20 medicine journals in 2008 as ranked by SCImago Journal Rank, five clearly align with IG's mandate: Nature Genetics, Development, PLoS Genetics, DNA Repair and Stem Cells. The collective percentage of Canadian content for these publications is 6.1%, compared to an average 5.3% for the top 20 publications and 3.5% for all publications ranked by SCImago. Thus, Canadian researchers in IG-related areas are publishing at disproportionately high levels in top-ranked journals.
The following are examples of high-profile publications produced by CIHR-funded researchers in IG-related areas in 2008:
-
With collaborators in France and Germany, Dr. Helen McNeill and her University of Toronto colleagues showed that an intercellular signaling pathway first characterized in fruit flies is conserved in mice and is required for kidney development.Footnote 2 Loss of this gene results in cystic kidney disease.
-
In the December 2008 issue of Nature Genetics, Dr. Eric Shoubridge of McGill University reported on how genes can mutate as they move from mother to offspring, allowing hereditary diseases to take hold.Footnote 3 The findings, based on mouse models, could help shed light on how stroke, epilepsy, diabetes, deafness and blindness get passed on.
-
A Toronto-based study led by Dr. Corey Nislow used yeast to screen a battery of widely prescribed psychoactive drugs for off-target effects. They found that more than a third of such drugs inhibited growth of normal yeast and affected multiple fundamental cellular processes to human life.Footnote 4 This work promises to provide a rational and low-cost method for understanding the side effects of pharmaceuticals and minimizing their number and severity.
Ensuring that competitiveness is maintained
The IG Bridging Operating Grants provide one year of support to accomplished investigators who would otherwise experience an interruption or delay in research funding. The grants ensure that highly trained staffers remain in place. This program can also fast-track junior researchers into establishing a laboratory. Since the launch of the program IG has supported 155 projects, almost all receiving an excellent rating by the peer review panel. This program represents an investment of more than $12 million since 2002.
To evaluate the program, 72 nominated PIs from seven IG Bridging Operating Grant competitions were surveyed with a response rate of 85% (n=61/ 72). The resulting data make a compelling case that the program plays a critical role in continuing robust research programs and competitiveness across Canada. Ninety percent of respondents indicated they received subsequent funding from CIHR or another funding organization that depended on research supported by their IG grant. Moreover, 85% of respondents (n=52/61) felt they would be unable to sustain the momentum of their research project without bridge funding. Most remaining respondents would have relied on start-up funds or significantly reduced both staff and the time that they could have committed towards their projects.
Capacity building
At its inception, IG recognized that Canada lacked sufficient research capacity in several key areas and developed strategic programs to strengthen these communities. These efforts have been very successful, as described below.
The Collaborative Health Research Projects program, an interdisciplinary, cross-council program with the Natural Sciences and Engineering Research Council (NSERC), has attracted increasing application pressure and interest since CIHR joined it in 2004. In 2000, NSERC received 63 full applications, rising to 111 in 2004 and to 326 letters of intent (LOI) in 2010 (application pressure necessitated institution of an LOI phase). These numbers suggest the program is encouraging a growing community of researchers who work at the important interface between the physical and applied science and biomedical research. A priority for IG is to direct more funds into this program.
It is recognized that Canada lacks clinical genetics researchers, as is the case in many countries. In 2008, IG examined the success rates of clinical genetics research projects in the CIHR Open Operating Grants program from 2003–2007. Although success rates indicated that clinical genetics applications fared no worse than grants overall, it was clear that the clinical genetics research community was small. For example, out of thousands of applications submitted during this period, only 66 included nominated principal applicants with an MD working in clinical genetics research.
The IG Clinical Investigatorship program was instituted to address this critical shortage. Early evidence suggests that the program is achieving its desired impact. A recent survey of five former awardees confirmed that all were still active in research, had external research funding, had protected research time, and were publishing research papers (8 to 31 per awardee). Fifteen clinical investigators have been supported by IG. Evaluation efforts are underway to capture the results of additional awards where the term has been completed.
Dr. Jacques Michaud, a former IG Clinical Investigatorship awardee, and his colleagues at Sainte-Justine Hospital in Montreal have successfully identified genes associated with mental retardation. In findings published in the New England Journal of Medicine and supported by an IG Genomic Medicine and Human Development Operating Grant, Dr. Michaud reported that a small but significant fraction of mentally retarded patients have genetic defects in a gene for the protein SYNGAP1, found in synapses, the specialized sites of communication between neurons.Footnote 5 Identification of the genetic causes of mental retardation helps affected families cope with the disorder and provides the first step in developing effective treatments.
Canada lacked a cadre of researchers with the analytic expertise to bridge laboratory-based to population-based research in 2000–2001. At that time, CIHR was investing $2.6 million under the Open Operating Grant Program and $577,000 under open personnel support programs. The Institute's efforts to develop the population genetics and genetic epidemiology community in partnership with IPPH (such as through directed strategic initiatives and by establishing a national meeting) contributed to its growth. By 2009–2010, $8.5 million was being invested by CIHR under the grant programs (both open and strategic) with $6.6 million funded under the open competitions. Also, the number of training awards funded increased from 10 in 2000–2001 to 41 in 2009–2010, primarily in the open competitions.
In 2001, there were few Canadian investigators in the area of Health Services for Genetic Diseases as evidenced by the complete lack of CIHR operating grant, salary or training investments in that year. By 2005, a new, cross-disciplinary, collaborative research community had taken root and CIHR was investing almost $2 million in strategic operating grants, primarily emerging teams and development grants funded by IG and IHSPR. By 2009–2010, a small but strong research community had been established in Canada and CIHR was investing more than $4.7 million in operating grant support, including $2.4 million under the CIHR Open Grant program. Moreover, 12 training awards were being funded with more awarded under the CIHR open competitions. Overall, CIHR funding went from approximately zero in 2000–2001 to $5.2 million in 2009–2010.
CIHR funding in support of genetic, ethical, legal and social issues (GELS) research amounted to less than $200,000 in 2000–2001. Investment in operating grants peaked at $1.63 million in 2007–2008 and declined to $943,000 in 2009–2010 (of which $600,000 is being supported by strategic funding). Unlike the population genetics community, this group of researchers has had difficulty transitioning from directed strategic funding to the open competition. However, there has been a steady rise in the number of training awards, from zero in 2000–2001, to six in 2005–2006, and to 10 in 2009–2010, which suggests that capacity-building efforts are having some success. The Institute remains committed to exploring how it can help foster additional activity and prominence for Canadian researchers in these important areas by supporting investigator-initiated and independent research. Part of IG's role is to support the basic and applied genetic ethical, legal and social issues (GELS) community, where other funders of GELS research emphasize a more applied service role.
Informing decision making
Two examples of translational impacts that resulted from the work of the IG GELS Priority and Planning Committee have to do with genetic discrimination and direct-to-consumer testing. New technologies such as whole genome sequencing and the development of new genetic tests create the potential for misuse or misinterpretation of genetic information. To address this issue, we brought together a group of experts in 2009 to identify areas of consensus for policy development. This resulted in an article entitled Keeping the GINA in the bottle: assessing the current need for genetic non-discrimination legislation in Canada.Footnote 6 In addition, the initiative led to a partnership with Genome Canada, which, in turn, resulted in a policy brief that synthesized academic knowledge on the topic and translated it into a form familiar to senior federal policy makers.
Direct-to-consumer (DTC) genetic testing activities have expanded dramatically in recent years, drawing attention from regulators, professional groups and the media. Outside Canada, government, professional and consumer groups have issued reports and position statements on DTC genetic testing. However, there has been little such activity in Canada. To advance discussion of DTC genetic tests in a Canadian context, the IG organized an expert forum to consider the current state of science, business and regulatory activities, public awareness and social and ethical issues. Recommendations for communication, educational, policy and regulatory tools were published in an article entitled Direct-to-consumer genetic testing; good, bad or benign? in Clinical Genetics.Footnote 7
An exciting initiative involving active participation of decision makers in Quebec was APOGÉE-NET. This IG- and IHSPR-funded knowledge network led by Dr. Renaldo Battista (Université de Montréal) demonstrated that a network involving researchers, decision makers, clinicians and citizens was an effective mechanism for addressing complex issues related to translation of genetic and genomics research. Resulting publications include "Genetic Screening: A conceptual model for programmes and policy making"Footnote 8, "Reconsidering reproductive benefit: A systematic review of guidelines on preconception, prenatal and newborn screening"Footnote 9, and "Guiding policy and decisions for genetic screening: Developing a systematic and transparent approach".Footnote 10
In 2005, IG and IHSPR funded CanGèneTest, an interdisciplinary research team that studied the translation of genetic laboratory services from discovery to clinical use. In 2009, the CanGèneTest consortium merged with APOGÉE-Net to form a new emerging team called the APOGÉE-Net/CanGèneTest Research and Knowledge Network on Genetic Services, funded by IG and IHSPR until 2014. The team, led by François Rousseau (Université Laval), is composed of 29 researchers, six decision makers and collaborators from Canada and abroad, and covers the whole spectrum of research expertise needed to validate and transfer genetic innovations into the health care system. It involves six Canadian universities, and has links with several Canadian and international research projects, as well as with Health Canada and with the ministries of health of Quebec and Nova Scotia.
Health and health system/care impacts
Following are two examples that demonstrate the impact of IG-supported research on the health care system.
Economic evaluations of health interventions
These are an important component of evidence-based decision making in clinical and public health settings or contexts. In the latter, economic evaluation can be especially complicated as options are numerous and outcomes can be measured only after an extended period of time. The CanGèneTest research consortium is investigating how computer simulations might help lead to rational decision making. Findings from initial simulations are already in press.Footnote 11,Footnote 12
Family physicians
These health care professionals are willing to play a significant role in delivering genetics services but are challenged by lack of knowledge about genetics and the rapid pace of genetic discovery. IG and IHSPR funded a multidisciplinary project led by Dr. June Carroll (Mt. Sinai Hospital, University of Toronto) that investigated whether a multi-faceted knowledge translation intervention would improve skills in primary care genetics. The intervention consisted of an interactive educational workshop, a portfolio of practical clinical genetics tools and an innovative knowledge support service called Gene Messenger. When presented with 10 clinical vignettes, family physicians who received the intervention were significantly more likely to make appropriate decisions whether or not to refer to genetics. They also had significantly increased overall self-reported confidence on core genetics competencies. Based on these results, the College of Family Physicians of Canada agreed to distribute Gene Messenger to its members.
Economic impacts
IG-funded research at the interface between the physical and applied sciences is starting to translate into substantial economic impact. Examples follow.
Dr. Carl Hansen, a CIHR New Investigator and Michael Smith Scholar, was recruited back to the University of British Columbia from the California Institute of Technology. He is funded by both an RMNI-IG Team Grant and an Invention: Tools, Techniques and Devices grant. His invention grant is on applying megapixel polymerase chain reaction (PCR) to non-invasive prenatal diagnostics. Using highly scalable microfluidics, Dr. Hansen's group has developed digital PCR technologies that achieve much higher levels of sensitivity, specificity and precision for nucleic acid analysis. They are able to separate samples into millions or billions of picolitre volume reactors, and amplify and detect isolated single molecules for the digital quantification of molecular concentration. Beyond pre-natal diagnostics, this technology could have wide application in diverse fields such as aging research, cancer biology, virology and clinical diagnostics.
Although modern optical imaging techniques can provide unprecedented details about life on a microscopic level, most of these techniques rely on the introduction of fluorescent probes that may perturb and alter the system. Label-free imaging is not only desirable, but may be necessary in many biomedical applications. A team funded by RMNI and IG developed the use of coherent anti-Stokes Raman scattering (CARS), a label-free, chemically specific imaging technique. There was no existing CARS hardware suitable for biomedical imaging, so this team developed an add-on CARS module that could be used with a commonly available multiphoton microscope system sold by Olympus.
Olympus had not succeeded in adding this functionality to its own microscope. Following commercialization discussions, Olympus now sells this CIHR-funded National Research Council (NRC) module and has opened a CARSLab Microscopy Facility at the NRC Steacie Institute to provide training for the biomedical research community. This project is a significant advance for the NRC Steacie Institute as it is their first successful in-house commercialized product.
Transformative effects of the Institute
Based on the evidence discussed above, IG transformed the Canadian health research enterprise by:
- advancing the integration of physical scientists and engineers into biomedical research
- increasing clinical genetics researcher capacity in Canada
- cultivating increased leadership capacity in population genetics and genetic epidemiology
- fostering the growth of a strong group of researchers in health services from a very limited base
- providing continued support to build the GELS research community
Additionally, and very importantly, IG has presided over a noteworthy increase in both the quality and quantity of Canadian research papers in genetics, basic biochemistry and cell biology. In all of its efforts, the IG works with the research community and partners to identify and respond to national and international opportunities and needs, and has engaged the community in the implementation and oversight of our activities. There are numerous additional examples of activities that would not have been possible without IG, including the following examples.
Catalyzing the Clinical Investigator Trainee Association of Canada
The Institute played a catalytic role in the formation of the Clinical Investigator Trainee Association of Canada (CITAC). Membership includes trainees in combined MD/PhD programs (125), clinical investigation programs (35), and MD/MSc programs (28). In addition to organizing a national meeting, CITAC has a governance structure, a trainee database, a website and a mentorship program.
Working with the voluntary health organizations in real time
The Institute's partnership activities have been greatly facilitated by the Canadian Genetic Coalition, the former IG Voluntary Health Organizations (VHO) Working Group. For example, when IG was designing the From Genes to Proteins, Cells, Tissues and Patients funding opportunity, the coalition actively participated in a consultative workshop and orchestrated a follow-up meeting with more than 15 VHOs, including the Canadian Organization for Rare Disorders (which represents more than 45 VHOs). Another example of the coalition's work is the health charities environmental scan. Information from 50 health charities is now being sought regarding how much financial and non-financial support they provide for research, including what research areas are supported and what gaps exist.
Workshops with impact and uptake
An example is the Personal Genome Workshop: Emerging Ethical, Legal and Social Challenges (November 2007), which represented one of the first transnational efforts to develop consensus guidelines for whole genome sequencing. The workshop recommendations were subsequently published in PLoS Biology and are frequently used worldwide.Footnote 13
Engaging the public
Over the past 10 years, public outreach has been an area of focus for IG. For example, the Institute organized Café Scientifiques that provided insight into health-related issues of popular interest. The Institute also partnered with the CIHR Marketing and Communications Branch to co-host the first-ever CIHR Science Writers Workshop that brought Canada's top medical reporters and senior communicators from the VHO community together with renowned researchers in the field of genetics.
Strengthening knowledge to action
As an example of its knowledge translation efforts, the IG's partnership with the CIHR Knowledge Synthesis and Exchange Branch in June 2007 brought together for an interactive workshop 20 leaders with a diverse range of experiences in translating research findings. Participants included Drs. Stanley Zlotkin (Hospital for Sick Children), Michael Hayden (University of British Columbia) and Jeremy Grimshaw (University of Ottawa). The meeting recommendations have been a valuable planning and program development resource for the Institute.
Going Forward
Transitioning to Roadmap
As IG embarks on a strategic planning process, it is understood that the goals of CIHR's strategic plan will influence and shape the future research priorities of the Institute. In an important partnership with CIHR, Genome Canada has launched a call for proposals under the Advancing Technology Innovation through Discovery strategic initiative. Rare diseases and pediatric cancers are the two priority areas for this initiative. The IG played an essential role in developing this initiative with ICR and Genome Canada, plus catalyzing a consortium of clinical geneticists working in the area of rare diseases that would be well positioned to respond to the funding opportunity. This first initiative is short-term in nature, but IG and Genome Canada view it as a pilot project that, if successful, will lead to a deeper collaboration between the organizations. The Institute is gratified by this, as it fulfills a recommendation from the 2006 International Review Report for improved clarity about the relationship.
Longer-term funding for projects concerning rare diseases will be possible through a related initiative called Rare Diseases Emerging Teams: Translating Basic Biology to Enhanced Patient Care, which is being launched by IG and the Institute of Nutrition, Metabolism and Diabetes in partnership with the Institute of Cancer Research, the Institute of Gender and Health, the Institute of Health Services and Policy Research, the Institute of Musculoskeletal Health and Arthritis, Ataxia of Charlevoix-Saguenay Foundation, Canadian Cystic Fibrosis Foundation, Canadian Organization for Rare Disorders, the Kidney Foundation of Canada and the Foundation Fighting Blindness, Canada in October 2010. Importantly, this $14.5 million strategic initiative, which was in an advanced stage of development when the Genome Canada opportunity arose, will fund two types of teams, one focused on basic biology, clinical research, and/or population research, and a second concerned with translational health services research, policy, economics and/or ethical, legal and social issues.
Often, these diseases have been relatively neglected in research and the coordination of service delivery, yet advances in genomic technologies and reductions in their costs are now allowing rapid progress toward identifying the responsible genes, which, in turn, have enhanced the availability of diagnostics and therapeutics. Moreover, expertise in animal model studies, clinical genetics, and population analysis can be brought to bear on understanding the functions of these genes and how mutations in them can cause disease, while health services and policy research can address issues in the regulation, appraisal and delivery of effective services.
Implementing Roadmap
To better link with Roadmap, the Rare Diseases initiative will soon be merged into a larger one centering on personalized medicine in which ICR also plays a leading role. Details of the personalized medicine initiative are still being developed, but one of its main objectives, aligning with those of the Rare Diseases initiative, includes improving, discovering, validating, evaluating and clinically applying new and existing biomarkers and genomic signatures for major chronic diseases that lead to patient stratification for effective treatment.
The other major Roadmap initiative that IG is co-leading, this time with the Institute of Neurosciences, Mental Health and Addiction (INMHA), is directed at complex diseases that likely involve the interplay between specific genes and environmental factors related to epigenetic regulation. We believe that this emerging area of health research holds great promise of deep insights that will promote health and reduce the burden of chronic disease and mental illness. Funding provided by IG, INMHA and partners to this strategic initiative will allow Canada to accept an invitation for full membership in the International Human Epigenome Consortium. We also anticipate that this initiative will deepen the links we are building with Genome Canada, whose Science and Technology Innovation Centres are prime facilities for the determination of epigenetic data. Future directions anticipated by the Structural Genomics Consortium (described in the previous section) are also within the realm of this initiative.
Strengthening international efforts
Health research is an endeavour that transcends national boundaries, and bibliometric analysis suggests that research articles co-authored by international teams of investigators have a higher average impact than those that originate from a single country. The Institute is committed to fostering an international consortium led by Canadian researchers. One important example is the Structural Genomics Consortium, which was discussed in an earlier section of this report.
The Institute is also playing an important role in the Autism Genome Project, led by Dr. Stephen Scherer (Hospital for Sick Children, Toronto). This major international initiative brings together many of the leading geneticists, clinicians and genome scientists undertaking autism research in Canada, and who link to 170 scientists from 10 other countries worldwide. This project is screening the genomes from more than 6,000 members of 1,600 families to identify susceptibility genes. The project is also incorporating genetic information about autism into health care delivery and policy development, which will eventually lead to new and more accurate diagnostic tests. In partnership with Autism Speaks, a voluntary health organization relevant to this area, Medical Research Council–UK, and INMHA, IG is working to determine future mechanisms of support as this project enters its third phase.
Additionally, IG is becoming a partner in the Genomics and Genetic Epidemiology of Multifactorial Disease (GenERA) network. The GenERA initiative is being launched in partnership with the health research agencies of Germany, Spain and France, and with the Fonds de la recherche en santé du Québec. The main goal of GenERA is to design and implement a series of funding intended to improve the utilization of genomic and proteomic data in clinical situations. IG also intends to raise its international profile through smaller-scale bilateral exchanges and collaboration arrangements.
Addressing future challenges
The Institute and the community it represents will face five main challenges in the coming years, including:
-
maintaining and improving the present positive trends in the quantity and quality of Canadian research relevant to the IG mandate, in the likely context of a challenging overall funding environment and at a time when research in the area is becoming increasingly reliant on large-scale approaches that require complex collaborations and substantial investment
-
striving to enable Canadian researchers with leading roles in international projects to command sufficient resources to carry out those roles
-
ensuring that our successes in fundamental research are efficiently and effectively translated to the clinic, while taking due caution not to encourage overly accelerated translation, which can result in misinformation and actual harm to patients and their caregivers
-
ensuring that funding for large-scale experimentation involving genomics, proteomics and other high-throughput technologies is available as support from numerous sources, often based in multiple countries, is normally required
-
fostering a robust discussion of ethical and policy questions concerning the use and misuse of genetic testing information in the particular context of the Canadian health care system
For the final challenge, inexpensive sequencing and proteomics-based methods that are coming soon are spawning a testing industry that has the potential to revolutionize health care. These developments also bring with them serious ethical questions concerning their appropriate use and the appropriate uses of the data that result. The Institute must foster research and discussions that help ensure that Canada does not rely solely on guidelines developed elsewhere concerning these matters, but instead that the best solutions are found in the context of, and for, Canadian society.
Conclusion
In its first 10 years, IG has established itself as CIHR's chief advocate for fundamental research in genetics, cell biology and biochemistry, and its activities have been associated with increased Canadian research productivity in all these areas. The Institute has also acted effectively with regard to its mandate in the other three CIHR themes. It has fostered successful new programs to advance clinical and population-based research, and partnered with other institutes toward progress in health services research and with regard to ethical, legal and social issues.
In the spirit of Roadmap, and in the context of the ever-increasing trend toward large-scale projects in relevant research areas, IG is developing new, more comprehensive programs aimed at further strengthening Canadian biomedical research. To do this, IG must continue to partner not only within CIHR and with its dedicated voluntary health organization community, but also with potential international partners who can be interested in supporting research teams in which Canadians are prominent players. The next five years promise to be exciting – even revolutionary – in the arena of genetics. The Institute is committed to ensuring that Canada is a world leader both in producing the new genetic revolution and in the application of genetics to the benefit of humankind.
List of Acronyms and Abbreviations
CIHR Institutes
| IAPH | Institute of Aboriginal Peoples' Health |
| IA | Institute of Aging |
| ICR | Institute of Cancer Research |
| ICRH | Institute of Circulatory and Respiratory Health |
| IGH | Institute of Gender and Health |
| IG | Institute of Genetics |
| IHSPR | Institute of Health Services and Policy Research |
| IHDCYH | Institute of Human Development, Child and Youth Health |
| III | Institute of Infection and Immunity |
| IMHA | Institute of Musculoskeletal Health and Arthritis |
| INMHA | Institute of Neurosciences, Mental Health and Addiction |
| INMD | Institute of Nutrition, Metabolism and Diabetes |
| IPPH | Institute of Population and Public Health |
IG Specific
| ARC | average of relative citations |
| CARS | coherent anti-Stokes Raman scattering |
| CGCF | Canadian Gene Cure Foundation |
| CITAC | Clinical Investigator Trainee Association of Canada |
| DTC | direct-to-consumer |
| GELS | genetic ethical, legal, and social issues |
| GenERA | Genomics and Genetic Epidemiology of Multifactorial Disease |
| GPS | Genomics, Public Policy and Society |
| HD | Huntington's Disease |
| IAB | Institute Advisory Board |
| KT | knowledge translation |
| LOI | letter of intent |
| MeSH | U.S. National Library of Medicine Medical Subject Headings |
| NRC | National Research Council |
| NSERC | Natural Sciences and Engineering Research Council |
| OST | Observatoire des Sciences et des Technologies |
| PCR | polymerase chain reaction |
| PI | principal investigator |
| RMNI | Regenerative Medicine and Nanomedicine Initiative |
| SCI | Science Convergence Initiative |
| SGC | Structural Genomics Consortium |
| SI | specialization index |
| STIHR | Strategic Training Initiative in Health Research |
| UK | United Kingdom |
| VHO | voluntary health organizations |
- Date modified: